
Chemistry Syllabus For The JEE Advanced 2023 Exam
Aspirants of JEE Advanced find Chemistry as an interesting subject to read. It is relatively easier to build concepts in Chemistry as compared to other subjects. When it comes to scoring, Chemistry proves to be a little tedious. To ensure students fetch a good score in the said subject in JEE Advanced 2023, here we provide detailed JEE Advanced 2023 Chemistry syllabus.
Complete JEE Advanced Syllabus Chemistry 2023
The Chemistry syllabus is broadly divided into 3 sections - Physical Chemistry, Inorganic Chemistry and Organic Chemistry.
Physical Chemistry –
|
Chapters |
Topics |
|
Surface Chemistry |
Colloids: types, methods of preparation, and general properties |
|
Elementary concepts of adsorption (excluding adsorption isotherms) |
|
|
Elementary ideas of emulsions, surfactants, and micelles (only definitions and examples) |
|
|
Nuclear Chemistry |
Radioactivity: isotopes and isobars |
|
Stability of nuclei with respect to proton-neutron ratio |
|
|
Properties of α, β and γ rays; Kinetics of radioactive decay (decay series excluded), carbon dating |
|
|
A brief discussion on fission and fusion reactions |
|
|
General Topics |
Concept of atoms and molecules |
|
Mole concept |
|
|
Dalton’s atomic theory |
|
|
Chemical formulae |
|
|
Calculations (based on the mole concept) involving common oxidation-reduction, neutralization, and displacement reactions |
|
|
Concentration in terms of mole fraction, molarity, molality and normality |
|
|
Balanced chemical equations |
|
|
Gaseous and Liquid States |
Kinetic theory of gases, average, root mean square and most probable velocities and their relation with temperature |
|
Absolute scale of temperature, ideal gas equation; Deviation from ideality, van der Waals equation |
|
|
Vapour pressure |
|
|
Law of partial pressures |
|
|
Diffusion of gases |
|
|
Atomic Structure and Chemical Bonding |
Wave-particle duality, de Broglie hypothesis |
|
Bohr model, spectrum of hydrogen atom, quantum numbers |
|
|
Uncertainty principle |
|
|
Aufbau principle |
|
|
Electronic configurations of elements (up to atomic number 36) |
|
|
Qualitative quantum mechanical picture of hydrogen atom, shapes of s, p and d orbitals |
|
|
Hybridisation involving s, p and d orbitals only |
|
|
Pauli’s exclusion principle and Hund’s rule |
|
|
Hydrogen bond |
|
|
Orbital overlap and covalent bond |
|
|
VSEPR model and shapes of molecules (linear, angular, triangular, square planar, pyramidal, square pyramidal, trigonalbipyramidal, tetrahedral and octahedral) |
|
|
Orbital energy diagrams for homonuclear diatomic species |
|
|
Polarity in molecules, dipole moment (qualitative aspects only) |
|
|
Energetics |
Internal energy, work and heat, pressure-volume work |
|
First law of thermodynamics |
|
|
Second law of thermodynamics |
|
|
Enthalpy, Hess’s law |
|
|
Entropy |
|
|
Heat of reaction, fusion and vapourization |
|
|
Criterion of spontaneity |
|
|
Free energy |
|
|
Chemistry Equilibrium |
Law of mass action |
|
Solubility product, common ion effect, pH and buffer solutions |
|
|
Equilibrium constant, Le Chatelier’s principle (effect of concentration, temperature and pressure |
|
|
Hydrolysis of salts |
|
|
Significance of ΔG and ΔG0in chemical equilibrium |
|
|
|
Acids and bases (Bronsted and Lewis concepts) |
|
Electrochemistry |
Standard electrode potentials |
|
Electrochemical cells and cell reactions |
|
|
Electrochemical series, emfof galvanic cells |
|
|
Nernst equation and its relation to ΔG |
|
|
Electrolytic conductance, specific, equivalent and molar conductivity, Kohlrausch’s law |
|
|
Faraday’s laws of electrolysis |
|
|
Concentration cells |
|
|
Chemical Kinetics |
Rate constant |
|
Rates of chemical reactions |
|
|
First order reactions |
|
|
Order of reactions |
|
|
Temperature dependence of rate constant (Arrhenius equation) |
|
|
Solid State |
Nearest neighbours, ionic radii, simple ionic compounds, point defects |
|
Classification of solids, crystalline state, seven crystal systems (cell parameters a, b, c, α, β, γ), close packed structure of solids (cubic), packing in fcc, bcc and hcp lattices |
|
|
Solutions |
Molecular weight determination from lowering of vapour pressure, elevation of boiling point and depression of freezing point |
|
Raoult’s law |
Scroll left or right to view full table
Inorganic Chemistry –
|
Chapters |
Topics |
|
Isolation/ Preparation and Properties (non-metals) |
Properties of allotropes of carbon (only diamond and graphite), phosphorus and sulphur |
|
Boron, silicon, nitrogen, phosphorus, oxygen, sulphur and halogens |
|
|
Preparation and Properties of the following compounds |
Boron: diborane, boric acid and borax |
|
Oxides, peroxides, hydroxides, carbonates, bicarbonates, chlorides and sulphates of sodium, potassium, magnesium and calcium |
|
|
Carbon: oxides and oxyacid (carbonic acid) |
|
|
Aluminium: alumina, aluminium chloride and alums |
|
|
Silicon: silicones, silicates and silicon carbide |
|
|
Oxygen: ozone and hydrogen peroxide |
|
|
Phosphorus: oxides, oxyacids (phosphorus acid, phosphoric acid) and phosphine |
|
|
Nitrogen: oxides, oxyacids and ammonia |
|
|
Halogens: hydrohalic acids, oxides and oxyacids of chlorine, bleaching powder; Xenon fluorides |
|
|
|
Sulphur: hydrogen sulphide, oxides, sulphurous acid, sulphuric acid and sodium thiosulphate |
|
Transition Elements (3 D Series) |
Coordination compounds: nomenclature of mononuclear coordination compounds, cis-trans and ionisation isomerisms, hybridization and geometries of mononuclear coordination compounds (linear, tetrahedral, square planar and octahedral) |
|
Definition, general characteristics, oxidation states and their stabilities, colour (excluding the details of electronic transitions) and calculation of spin-only magnetic moment |
|
|
Preparation and Properties of the following Compounds |
Potassium permanganate, potassium dichromate, silver oxide, silver nitrate, silver thiosulphate |
|
Oxides and chlorides of tin and lead; Oxides, chlorides and sulphates of Fe2+, Cu2+and Zn2+ |
|
|
Ores and Minerals |
Commonly occurring ores and minerals of iron, copper, tin, lead, magnesium, aluminium, zinc and silver |
|
Principles of Qualitative Analysis |
Nitrate, halides (excluding fluoride), sulphate and sulphide |
|
Groups I to V (only Ag+, Hg2+, Cu2+, Pb2+, Bi3+, Fe3+, Cr3+, Al3+, Ca2+, Ba2+, Zn2+, Mn2+and Mg2+) |
|
|
Extractive Metallurgy |
Chemical principles and reactions only (industrial details excluded) |
|
Self reduction method (copper and lead) |
|
|
Cyanide process (silver and gold) |
|
|
Electrolytic reduction method (magnesium and aluminium) |
|
|
Carbon reduction method (iron and tin) |
Scroll left or right to view full table
Organic Chemistry –
|
Chapters |
Topics |
|
Concepts |
Hybridisation of carbon |
|
Structural and geometrical isomerism |
|
|
Conformations of ethane and butane (Newman projections) |
|
|
σ and π-bonds |
|
|
IUPAC nomenclature of simple organic compounds (only hydrocarbons, mono-functional and bi-functional compounds) |
|
|
Optical isomerism of compounds containing up to two asymmetric centres, (R,S and E,Z nomenclature excluded |
|
|
Shapes of simple organic molecules |
|
|
Determination of empirical and molecular formulae of simple compounds (only combustion method) |
|
|
Keto-enoltautomerism |
|
|
Inductive and resonance effects on acidity and basicity of organic acids and bases |
|
|
Resonance and hyperconjugation |
|
|
Hydrogen bonds: definition and their effects on physical properties of alcohols and carboxylic acids |
|
|
Reactive intermediates produced during homolytic and heterolytic bond cleavage |
|
|
Polarity and inductive effects in alkyl halides |
|
|
Formation, structure and stability of carbocations, carbanions and free radicals |
|
|
Preparation, Properties and Reactions of Alkanes |
Homologous series, physical properties of alkanes (melting points, boiling points and density) |
|
Preparation of alkanes by Wurtz reaction and decarboxylation reactions |
|
|
Combustion and halogenation of alkanes |
|
|
Preparation, Properties and Reactions of Alkenes and Alkynes |
Physical properties of alkenes and alkynes (boilingpoints, density and dipole moments) |
|
Reactions of alkenes with KMnO4and ozone |
|
|
Acidity of alkynes |
|
|
Reduction of alkenes and alkynes |
|
|
Acid catalysed hydration of alkenes and alkynes (excluding the stereochemistry of addition and elimination) |
|
|
Electrophilic addition reactions of alkenes with X2, HX, HOX and H2O (X=halogen) |
|
|
Preparation of alkenes and alkynes by elimination reactions |
|
|
Metal acetylides |
|
|
Addition reactions of alkynes |
|
|
Reactions of Benzene |
Electrophilic substitution reactions: halogenation, nitration, sulphonation, Friedel-Crafts alkylation and acylation |
|
Structure and aromaticity |
|
|
Effect of o-, m-and p-directing groups in monosubstituted benzenes |
|
|
Phenols |
Reimer-Tieman reaction, Kolbe reaction |
|
Acidity, electrophilic substitution reactions (halogenation, nitration and sulphonation) |
|
|
Characteristic Reactions of the following (including those mentioned above) |
Alcohols: esterification, dehydration and oxidation, reaction with sodium, phosphorus halides, ZnCl2/concentrated HCl, conversion of alcohols into aldehydes and ketones |
|
Alkyl halides: rearrangement reactions of alkyl carbocation, Grignard reactions, nucleophilic substitution reactions |
|
|
Aldol condensation, Perkin reaction |
|
|
Aldehydes and Ketones: oxidation, reduction, oxime and hydrazone formation |
|
|
Ethers: Preparation by Williamson’s Synthesis |
|
|
Amines: basicity of substituted anilines and aliphatic amines, preparation from nitro compounds, reaction with nitrous acid, azo coupling reaction of diazonium salts of aromatic amines, Sandmeyer and related reactions of diazonium salts |
|
|
Cannizzaro reaction |
|
|
Carbylamine reaction |
|
|
Haloform reaction and nucleophilic addition reactions (Grignard addition) |
|
|
Haloarenes: nucleophilic aromatic substitution in haloarenes and substituted haloarenes (excluding Benzyne mechanism and Cine substitution) |
|
|
Carboxylic acids: formation of esters, acid chlorides and amides, ester hydrolysis |
|
|
Carbohydrates |
Classification |
|
Oxidation, reduction, glycoside formation and hydrolysis of sucrose |
|
|
Mono-and di-saccharides (glucose and sucrose |
|
|
Amino Acids and Peptides |
General structure (only primary structure for peptides) and physical properties |
|
Practical Organic Chemistry |
Detection and identification of the following functional groups: hydroxyl (alcoholic and phenolic), carbonyl (aldehyde and ketone), carboxyl, amino and nitro |
|
Detection of elements (N, S, halogens) |
|
|
Chemical methods of separation of mono-functional organiccompounds from binary mixtures |
|
|
Properties and Uses of some important polymers |
Natural rubber, cellulose, nylon, teflon and PVC |
Scroll left or right to view full table
Section-wise distribution of marks in Chemistry of JEE (Advanced) 2022
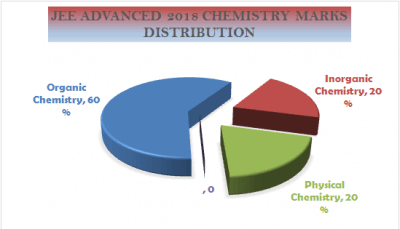
|
Name of the Section |
Approximate marks distribution |
|
Organic Chemistry |
45 |
|
Inorganic Chemistry |
35 |
|
Physical Chemistry |
40 |
Scroll left or right to view full table
Topic – Wise distribution of marks in the Chemistry Syllabus of JEE (Advanced) 2023
|
S. No. |
Name of the Section |
Name of the Topics |
Approximate marks per Topic |
|
1. |
Organic Chemistry |
Amines |
10 -11 |
|
General Organic Chemistry |
8-9 |
||
|
Bio-molecules |
3-4 |
||
|
Stereoisomerism |
5-6 |
||
|
Polymers |
7-8 |
||
|
Aromatic Compounds |
6-7 |
||
|
Carbonyl Compounds |
3 -4 |
||
|
Total |
45 marks. |
||
|
2. |
Inorganic Chemistry |
P- Block Elements |
11 |
|
Chemical Bonding |
08 |
||
|
Metallurgy |
04 |
||
|
Co-ordination Compounds |
06 |
||
|
Qualitative Analysis |
08 |
||
|
Total |
36 |
||
|
3. |
Physical Chemistry |
Chemical Equilibrium |
5- 6 |
|
Gaseous State |
2-3 |
||
|
Chemical Kinetics |
8-9 |
||
|
Mole Concept |
8-9 |
||
|
Surface Chemistry |
2-3 |
||
|
Solid State |
4-5 |
||
|
Solution and Colligative Properties |
3-4 |
||
|
Atomic Structure and Nuclear Chemistry |
2-3 |
||
|
Electrochemistry |
3-4 |
||
|
Total |
43 |
Scroll left or right to view full table
Some of the most important topics with marks weightage of around 10% are – Amines, General Organic Chemistry, Polymers, P-Block elements, Qualitative Analysis, Mole concept, and Chemical Kinetics.

0 Comments